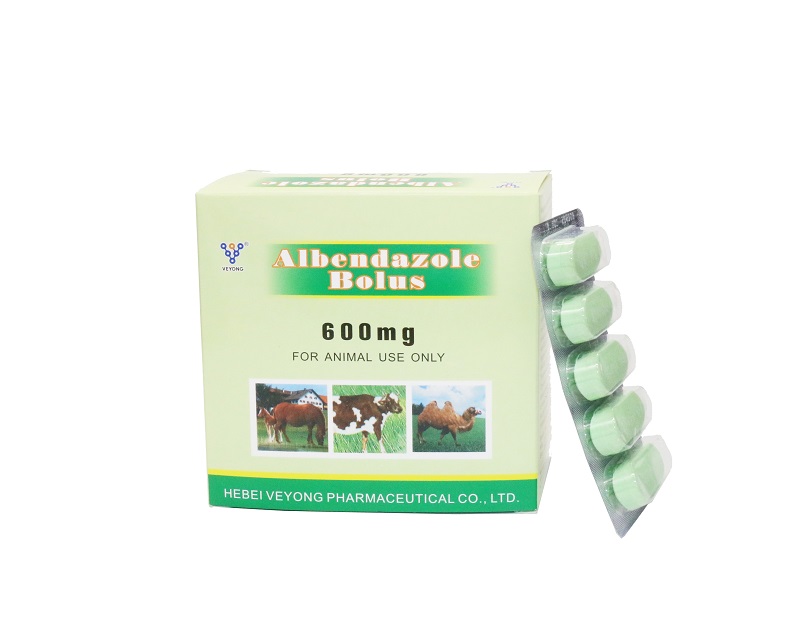
600mg Albendazole bolus for Cattle
Composition
Each bolus contains: Albendazole 600mg
Description
Albendazole has a broad spectrum of anthelmintic action, is active against nematodes, trematodes and cestodes. Albendazole is rapidly absorbed from the gastrointestinal tract. The maximum concentration of albendazole in the blood serum is observed 12 to 25 hours after the drug is used.
The mechanism of action of albendazole is fueled by metabolic disorders, inhibition of fumarate-reductase activity and parasite synthesis, which leads to the death of helminths. It is also an ovicide that helps reduce environmental pollution.
Indications
For deworming cattle, goats and sheep with the following diseases:
- gastrointestinal nematodes: Naetopshus, Ostertagia, Trichostrongylus, Strongyloides, Bunostomum, Cooperia, Nematodirus, Chabertia, Oes ophagostomum, Toxocara;
- pulmonary strongylids (mature and larval forms): Dictyocaulus, Muellerius, Protostrongylus, Neostrongylus, Cystocaulus;
- cestodes (scolexes and segments):
- adult flat worms: Fasciola spp. , Dicrocoelium lanceolatum
Dosage and administration
For orally.
Cattle: 1 bolus per 60kg live weight (10 mg albendazole per 1 kg live weight)
The usual dosage is 1/4 bolus per 30 kg body weight (5 mg albendazole per kg body weight). In case of infection with adult flatworms and protostrongylosis, the dose is increased to 1/2 bolus per 40 weight (7.5 mg of albendazole per 1 kg of live weight).
Contraindications
Do not use pregnant cows, and pregnant sheep and goats in the first half of pregnancy, as well as malnourished and sick animals.
Slaughter of animals for meat after deworming is allowed no earlier than after 27 days.
Milk-dairy animals must not be used for food purposes for 7 days after worming.
Side effects
At the recommended dosage, Albendazole has no significant adverse effects.
Withdrawal Time
27days before slaughter.
Not for use in dairy cattle of breeding age, or in any cattle during the first 45 days of pregnancy
Storage
Store in its original packaging at a temperature not exceeding 30 ° C.
Keep out of the reach of children .
Shelf life
3 years






Hebei Veyong pharmaceutical Co., Ltd, was established in 2002, located in Shijiazhuang City, Hebei Province, China, next to the Capital Beijing. She is a large GMP-certified veterinary drug enterprise, with R&D, production and sales of veterinary APIs, preparations, premixed feeds and feed additives. As Provincial Technical Center, Veyong has established an innovated R&D system for new veterinary drug, and is the nationally known technological innovation based veterinary enterprise, there are 65 technical professionals. Veyong has two production bases: Shijiazhuang and Ordos, of which the Shijiazhuang base covers an area of 78,706 m2, with 13 API products including Ivermectin, Eprinomectin, Tiamulin Fumarate, Oxytetracycline hydrochloride ects, and 11 preparation production lines including injection, oral solution, powder, premix, bolus, pesticides and disinfectant, ects. Veyong provides APIs, more than 100 own- label preparations, and OEM & ODM service.
Veyong attaches great importance to the management of EHS(Environment, Health& Safety) system, and obtained the ISO14001 and OHSAS18001 certificates. Veyong has been listed in the strategic emerging industrial enterprises in Hebei Province and can ensure the continuous supply of products.

Veyong established the complete quality management system, obtained the ISO9001 certificate, China GMP certificate, Australia APVMA GMP certificate, Ethiopia GMP certificate, Ivermectin CEP certificate, and passed US FDA inspection. Veyong has professional team of registeration, sales and technical service, our company has gained reliance and support from numerous customers by excellent product quality, high-quality pre-sales and after-sales service, serious and scientific management. Veyong has made long term cooperation with many internationally known animal pharmaceutical enterprises with products exported to the Europe, South America, Middle East, Africa, Asia, etc. more than 60 countries and regions.





.png)
.png)
.png)
.png)




