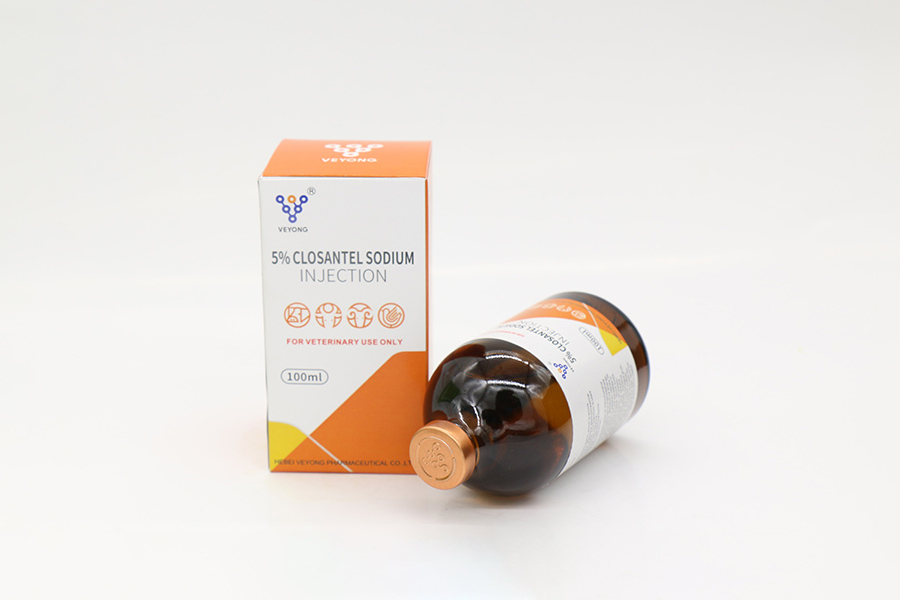
5% Closantel Sodium Injection
Pharmacological Action
Pharmacodynamics This product has anthelmintic activity against cattle, Fasciola lamblia, Haemonchus contortus and some arthropods. It is ineffective against Anterior and Posterior Onchocerca spp. For most gastrointestinal nematodes, such as Haemonchus, Yangkou, and Esophagostomum, more than 90%. Some H. contortus strains of sheep can develop resistance to this product. Closantel sodium is effective in expelling hookworms, but not larvae in the body. It also prevents or reduces Strongyloides vulgaris infection in horses. In addition, it has 100% killing effect on first, second and third stage sheep nose fly maggots, and also has good killing effect on third stage larvae of cow skin fly. Its anthelmintic mechanism is to exert an anthelmintic effect by increasing parasite mitochondrial permeability and uncoupling oxidative phosphorylation.
Pharmacokinetics
The plasma protein binding ofClosantel in sheep is high (> 99%), resulting in an elimination half-life of 14.5 Day. The drug was mainly excreted in the feces (80%), and less than 0.5% of the drug was excreted in the urine.
Drug Interactions
Closantel can be used in combination with benzimidazoles or levamisole.
Action and Use
Antihelminthic drugs. It is mainly used for the control of Fasciola hepatica, gastrointestinal nematode disease and sheep nasal myiasis in cattle and sheep.
Dosage and Administration
Subcutaneous or intramuscular injection: a single dose, 0.05 ~ 0.1ml for cattle and 0.1 ~ 0.2ml for sheep per 1 kg body weight.
Adverse Reactions
No adverse reactions have been observed when used according to the specified usage and dosage.
Precautions
It has certain irritation to local tissues.
Withdrawal Period
28 days for cattle and sheep; 28 days for milk withdrawal period.
Storage
Sealed and stored in a cool place, protected from light.
Package
50ml,100ml,250ml.
Hebei Veyong pharmaceutical Co., Ltd, was established in 2002, located in Shijiazhuang City, Hebei Province, China, next to the Capital Beijing. She is a large GMP-certified veterinary drug enterprise, with R&D, production and sales of veterinary APIs, preparations, premixed feeds and feed additives. As Provincial Technical Center, Veyong has established an innovated R&D system for new veterinary drug, and is the nationally known technological innovation based veterinary enterprise, there are 65 technical professionals. Veyong has two production bases: Shijiazhuang and Ordos, of which the Shijiazhuang base covers an area of 78,706 m2, with 13 API products including Ivermectin, Eprinomectin, Tiamulin Fumarate, Oxytetracycline hydrochloride ects, and 11 preparation production lines including injection, oral solution, powder, premix, bolus, pesticides and disinfectant, ects. Veyong provides APIs, more than 100 own- label preparations, and OEM & ODM service.
Veyong attaches great importance to the management of EHS(Environment, Health& Safety) system, and obtained the ISO14001 and OHSAS18001 certificates. Veyong has been listed in the strategic emerging industrial enterprises in Hebei Province and can ensure the continuous supply of products.

Veyong established the complete quality management system, obtained the ISO9001 certificate, China GMP certificate, Australia APVMA GMP certificate, Ethiopia GMP certificate, Ivermectin CEP certificate, and passed US FDA inspection. Veyong has professional team of registeration, sales and technical service, our company has gained reliance and support from numerous customers by excellent product quality, high-quality pre-sales and after-sales service, serious and scientific management. Veyong has made long term cooperation with many internationally known animal pharmaceutical enterprises with products exported to the Europe, South America, Middle East, Africa, Asia, etc. more than 60 countries and regions.





.png)
.png)
.png)
.png)