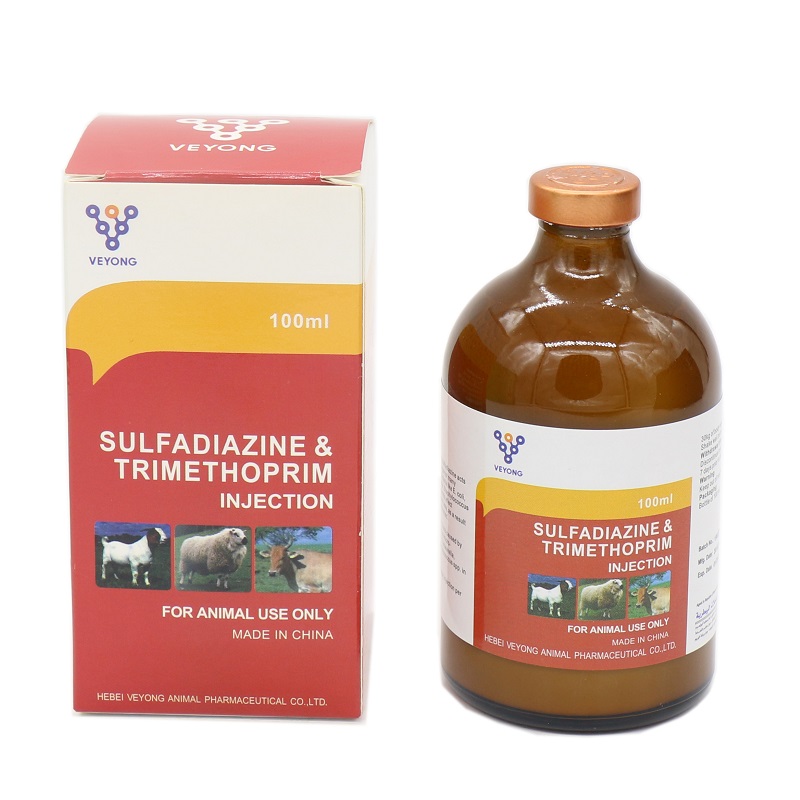
20% Sulfadiazine+4% Trimethoprim Injection
Active Ingredient
Sulfadiazine 20.00% w/v.
Trimethoprim 4.00% w/v
Pharmacological action
Sulfadiazine is a moderately effective sulfa drug for systemic use and is a broad-spectrum bacteriostatic agent. Its mechanism of action is because it is structurally similar to p-aminobenzoic acid (PABA) and can compete with PABA to act on the dihydrofolate synthase in bacteria, thereby preventing PABA from being used as a raw material to synthesize tetrahydrofolate required by bacteria, thereby inhibiting The synthesis of bacterial protein plays an antibacterial effect.
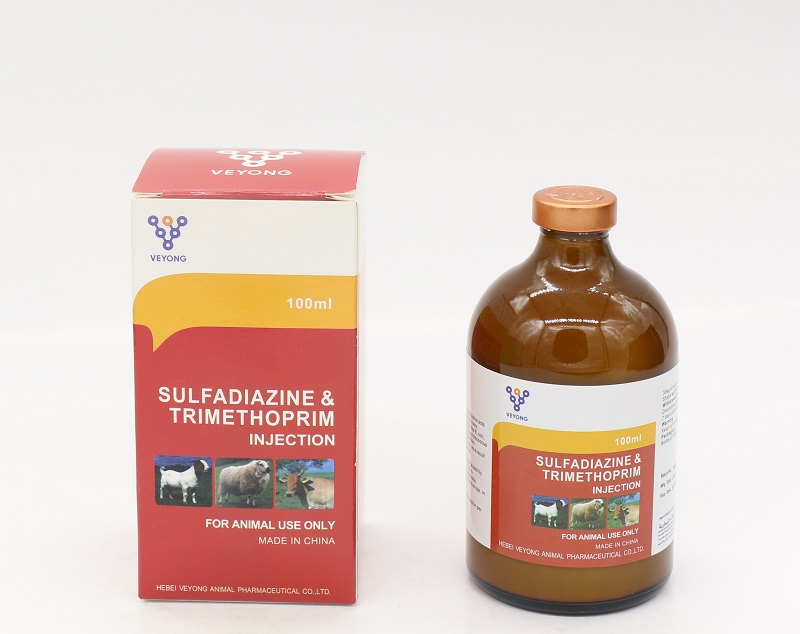
Indlcation
This Injectable Solution is indicated in the treatment of systemic infections caused by or associated with organisms sensitive to the Trimethoprim: Sulfadiazine combination. The spectrum of activity includes both Gram-positive and Gram-negative organisms including: Actinobacilli, Actinomycae, Bordetella spp, Brucella Corynebacteria, Escherichia coli, Haemophilus spp. Klebsiella spp, Pasteurella spp, Pneumococci. Proteus, Salmonella spp. Staphylococci, Streptococci, Vibrio.
Dosage and administration
By subcutaneous injection only.
Cattle: The recommended dose rate is 15 mg of active ingredients per kg body weight (1 ml per 16kg body weight) by intramuscular or slow intravenous injection.
Horses: The recommended dose rate is 15 mg of active ingredients per kg body weight (1 ml per 16 kg body weight), by slow intravenous injection.
Dogs and Cats: The recommended dose rate is 30mg of active ingredients per kg body weight (1 ml per 8 kg body weight ).
Contraindications
The injection should not be given by routes other than those recommended.
Not to be administered intraperitoneally, intra - arterially or intrathecally.
Do not administer to animals with known sulphonamide sensitivity, severe liver parenchymal damage or blood dyscrasias.
Special Warnings
1 For intravenous administration the product should be warmed to body temperature and injected slowly over as long a period as is reasonably practical.
2 At the first sign of intolerance the injection should be interrupted and shock treatment initiated.
Adequate drinking water should be available during the therapeutic effect of the product.
Withdrawal Period
Cattle : Meat - 12 days
Milk - 4 days.
Storage
Protect from direct sunlight and store below 30℃.
Hebei Veyong pharmaceutical Co., Ltd, was established in 2002, located in Shijiazhuang City, Hebei Province, China, next to the Capital Beijing. She is a large GMP-certified veterinary drug enterprise, with R&D, production and sales of veterinary APIs, preparations, premixed feeds and feed additives. As Provincial Technical Center, Veyong has established an innovated R&D system for new veterinary drug, and is the nationally known technological innovation based veterinary enterprise, there are 65 technical professionals. Veyong has two production bases: Shijiazhuang and Ordos, of which the Shijiazhuang base covers an area of 78,706 m2, with 13 API products including Ivermectin, Eprinomectin, Tiamulin Fumarate, Oxytetracycline hydrochloride ects, and 11 preparation production lines including injection, oral solution, powder, premix, bolus, pesticides and disinfectant, ects. Veyong provides APIs, more than 100 own- label preparations, and OEM & ODM service.
Veyong attaches great importance to the management of EHS(Environment, Health& Safety) system, and obtained the ISO14001 and OHSAS18001 certificates. Veyong has been listed in the strategic emerging industrial enterprises in Hebei Province and can ensure the continuous supply of products.

Veyong established the complete quality management system, obtained the ISO9001 certificate, China GMP certificate, Australia APVMA GMP certificate, Ethiopia GMP certificate, Ivermectin CEP certificate, and passed US FDA inspection. Veyong has professional team of registeration, sales and technical service, our company has gained reliance and support from numerous customers by excellent product quality, high-quality pre-sales and after-sales service, serious and scientific management. Veyong has made long term cooperation with many internationally known animal pharmaceutical enterprises with products exported to the Europe, South America, Middle East, Africa, Asia, etc. more than 60 countries and regions.




.png)
.png)
.png)
.png)